Air Quality Lab
Prelab Questions
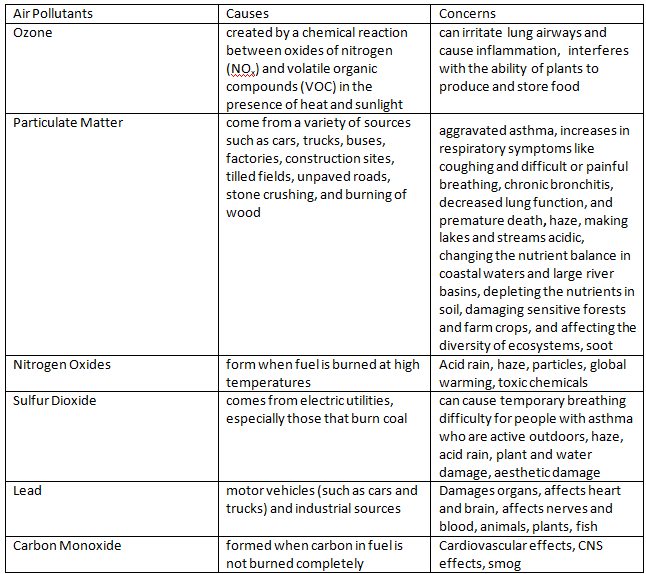
1. Create a chart showing the six air pollutants, causes, and concerns from the information in the pre-lab.
2. Why is ground level ozone highest in the summer and almost nonexistent in the winter? How might this affect our results as we test the air during this part of the year?
Ground level ozone is highest in the summer and almost nonexistent in the winter because of the difference in heat and sunlight. This might affect our results when we test the air because, as it’s winter, there will be less ground level ozone than if we had tested over the summer.
3. Many third world countries still use leaded gasoline (since they have older cars or since they have not forced changes). What concerns may the governments of these countries need to deal with?Lead poisoning can cause medical issues in the cardiovascular and nervous systems, which could lead to lawsuits if people afflicted by lead poisoning can pin it down to the car manufacturers.
4. Use the internet to find what parts of North Carolina are not in compliance with the Standards of the Clean Air Act. You may give either general areas or specific counties.
Plott Balsam Mountains – area above 4,000 feet in Haywood County; Great Balsam Mountains – areas above 4,000 feet in Haywood, Jackson and Transylvania regions; Great Smoky Mountains National Park – area in Haywood and Swain counties; Blue Ridge, Black and Great Craggy Mountains – areas above 4,000 feet in Buncombe, McDowell and Yancey counties; Asheville area, covering Buncombe County; Charlotte/Gastonia/Rock Hill area, including Gaston and Mecklenburg counties and portions of five other counties; Fayetteville area, covering Cumberland County; Greensboro/Winston-Salem/High Point area, including Alamance, Davidson, Forsyth, Guilford and portions of four other counties; Hickory/Morganton/Lenoir area, including portions of Burke, Caldwell, Catawba and Alexander counties; Raleigh/Durham/Chapel Hill area, including Durham, Orange, Wake and portions of five other counties; and the Rocky Mount area, including a portion of Edgecombe County
Ground level ozone is highest in the summer and almost nonexistent in the winter because of the difference in heat and sunlight. This might affect our results when we test the air because, as it’s winter, there will be less ground level ozone than if we had tested over the summer.
3. Many third world countries still use leaded gasoline (since they have older cars or since they have not forced changes). What concerns may the governments of these countries need to deal with?Lead poisoning can cause medical issues in the cardiovascular and nervous systems, which could lead to lawsuits if people afflicted by lead poisoning can pin it down to the car manufacturers.
4. Use the internet to find what parts of North Carolina are not in compliance with the Standards of the Clean Air Act. You may give either general areas or specific counties.
Plott Balsam Mountains – area above 4,000 feet in Haywood County; Great Balsam Mountains – areas above 4,000 feet in Haywood, Jackson and Transylvania regions; Great Smoky Mountains National Park – area in Haywood and Swain counties; Blue Ridge, Black and Great Craggy Mountains – areas above 4,000 feet in Buncombe, McDowell and Yancey counties; Asheville area, covering Buncombe County; Charlotte/Gastonia/Rock Hill area, including Gaston and Mecklenburg counties and portions of five other counties; Fayetteville area, covering Cumberland County; Greensboro/Winston-Salem/High Point area, including Alamance, Davidson, Forsyth, Guilford and portions of four other counties; Hickory/Morganton/Lenoir area, including portions of Burke, Caldwell, Catawba and Alexander counties; Raleigh/Durham/Chapel Hill area, including Durham, Orange, Wake and portions of five other counties; and the Rocky Mount area, including a portion of Edgecombe County
How much ground level ozone is there in the air outside Heritage High School compared to how much there is inside?
Hypothesis: There will be more ground level ozone in a more higher concentration outside of the school and less inside because outdoors the air is exposed to heat and light that helps the ozone to form. It is also exacerbated by exhaust from the parking lot, whereas there is less heat, light, and exhaust inside the school.
Conditions
Data collected 1/30/13-2/1/13
Weather: Blustery and very humid, drizzle
Temperature on 1/30/13: 20°C
Relative Humidity on 1/30/13: 74%
Parts of Experiment
Independent Variable: location of ozone monitoring test strips and particulate collectors
Dependent Variable: amount of ozone collected on strips and particulate collectors
Controlled Variables: size of strips, time strips and collectors are left out, substances on strips and collectors
Control Group: strip and collector left in closed Petri dish (Ms. Bostic's control)
Experimental Groups: strips and collectors left outside and in locations inside the school (Groups A-H)
Materials
Potassium Iodide
Corn starch
Beakers
Spatulas
Large Graduated Cylinder
Test Tube Rack
Filter Paper
Microscope slides
Petroleum Jelly
Q-tips
Microscopes
Procedure
1. Half the class should work on preparing the ozone monitoring test strips by placing 100ml of water in a 250 ml beaker, and adding 5g of cornstarch. Then heat and stir the mixture until it gels (when the mixture becomes thick and clear). Remove the solution from heat, add 1 g of potassium iodide, stir well, and allow to cool. After the solution has cooled, lay a piece of filter paper on a petri and brush the paste onto the filter paper. Turn the filter paper and brush the paste on the other side. Hang up and allow the filter paper to dry. Cut the papers into 1 inch-wide strips. Store the strips in a plastic bag out of sunlight.
2. The other half of the class will prepare Particulate matter collectors by smearing a thin layer of petroleum jelly onto a microscope slides. Try to make the layer as even as possible. Use a q-tip to lightly smear the specimen side of each slide. You will need to prepare 2 slides per group and place them in a slide container to avoid smearing the petroleum jelly on anyone.
3. Each group will place one labeled slide on the exterior of the school. Some groups should choose areas that would receive few particulates, others should choose locations that produce more particulates. (Keep in mind sources of particulate matter)
4. One member from each group should take the second slide and place it in a location inside the school.
5. You should then place your ozone monitoring strip outside for 8 hrs. Before placing the strip outside, dip the strip in distilled water, then attach the strip out of sunlight. When placing your strip find the relative humidity for that day.
6. The next day, each group should collect their slide and ozone monitoring strip.
7. Dunk each strip into distilled water. Use the chart at the right to determine the Schoenbein Number of your strip.
8. Use the relative humidity and the Schoenbein number to calculate the concentration of ground level ozone using the chart below.
9. Look at your particulate matter slide under the microscope. First decide the amount of particulates and then attempt to estimate the source of the PM. Your group will need to decide how you can classify what type of PM was collected.
10. Make sure you record all you lab information into your notebook.
Data collected 1/30/13-2/1/13
Weather: Blustery and very humid, drizzle
Temperature on 1/30/13: 20°C
Relative Humidity on 1/30/13: 74%
Parts of Experiment
Independent Variable: location of ozone monitoring test strips and particulate collectors
Dependent Variable: amount of ozone collected on strips and particulate collectors
Controlled Variables: size of strips, time strips and collectors are left out, substances on strips and collectors
Control Group: strip and collector left in closed Petri dish (Ms. Bostic's control)
Experimental Groups: strips and collectors left outside and in locations inside the school (Groups A-H)
Materials
Potassium Iodide
Corn starch
Beakers
Spatulas
Large Graduated Cylinder
Test Tube Rack
Filter Paper
Microscope slides
Petroleum Jelly
Q-tips
Microscopes
Procedure
1. Half the class should work on preparing the ozone monitoring test strips by placing 100ml of water in a 250 ml beaker, and adding 5g of cornstarch. Then heat and stir the mixture until it gels (when the mixture becomes thick and clear). Remove the solution from heat, add 1 g of potassium iodide, stir well, and allow to cool. After the solution has cooled, lay a piece of filter paper on a petri and brush the paste onto the filter paper. Turn the filter paper and brush the paste on the other side. Hang up and allow the filter paper to dry. Cut the papers into 1 inch-wide strips. Store the strips in a plastic bag out of sunlight.
2. The other half of the class will prepare Particulate matter collectors by smearing a thin layer of petroleum jelly onto a microscope slides. Try to make the layer as even as possible. Use a q-tip to lightly smear the specimen side of each slide. You will need to prepare 2 slides per group and place them in a slide container to avoid smearing the petroleum jelly on anyone.
3. Each group will place one labeled slide on the exterior of the school. Some groups should choose areas that would receive few particulates, others should choose locations that produce more particulates. (Keep in mind sources of particulate matter)
4. One member from each group should take the second slide and place it in a location inside the school.
5. You should then place your ozone monitoring strip outside for 8 hrs. Before placing the strip outside, dip the strip in distilled water, then attach the strip out of sunlight. When placing your strip find the relative humidity for that day.
6. The next day, each group should collect their slide and ozone monitoring strip.
7. Dunk each strip into distilled water. Use the chart at the right to determine the Schoenbein Number of your strip.
8. Use the relative humidity and the Schoenbein number to calculate the concentration of ground level ozone using the chart below.
9. Look at your particulate matter slide under the microscope. First decide the amount of particulates and then attempt to estimate the source of the PM. Your group will need to decide how you can classify what type of PM was collected.
10. Make sure you record all you lab information into your notebook.
Conclusion Questions
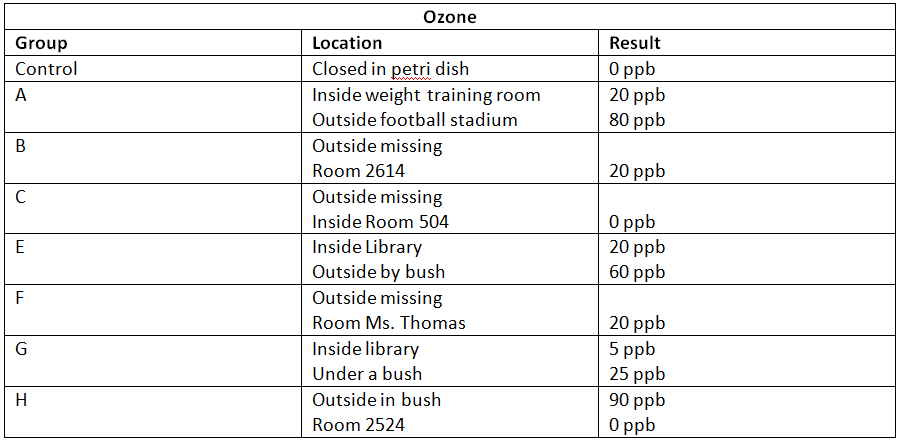
1. The highest rating the EPA will allow for a community for ground level ozone is 80 ppb. Any community with over 80 ppb for an average over three years is in noncompliance with the Clean Air Act. Compare your number to this standard. Did Wake Forest meet the standard on this day? Using what you know about the formation of ozone, decide if you believe the measurement you made was an accurate measurement of the air quality and why.
The number our group discerned from the experiment was 60, which is below the EPA's limit of 80. Wake Forest did meet the standard on January 31. I believe the measurement we made was mostly accurate because the slip was waving in open air, not directly in the area where all the exhaust fumes were so that the effect of the cars was indirect, as it would be for most people breathing. However, it was near a bush, which can tend to increase ground level ozone, so it's possible that that skewed the results a bit.
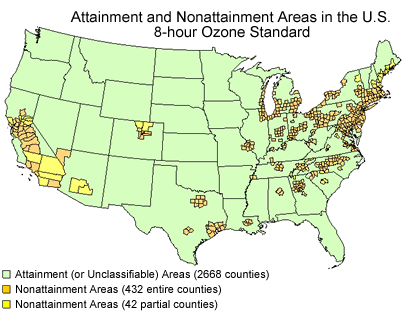
2. 13% of counties in the US are not meeting the Ozone requirements of the Clean Air Act. Use the chart below and information from class to explain the reason for the location of the counties in Non-attainment.
I think the reason the counties that are in non-attainment areas are there because of industrialism and population. Many of the densely populated big cities in the east of the country are non-attainment, while the more open country in the west has more attainment areas. Denver is an anomaly because it sits in a valley, so the ozone doesn't rise like it does in other places.
The number our group discerned from the experiment was 60, which is below the EPA's limit of 80. Wake Forest did meet the standard on January 31. I believe the measurement we made was mostly accurate because the slip was waving in open air, not directly in the area where all the exhaust fumes were so that the effect of the cars was indirect, as it would be for most people breathing. However, it was near a bush, which can tend to increase ground level ozone, so it's possible that that skewed the results a bit.
2. 13% of counties in the US are not meeting the Ozone requirements of the Clean Air Act. Use the chart below and information from class to explain the reason for the location of the counties in Non-attainment.
I think the reason the counties that are in non-attainment areas are there because of industrialism and population. Many of the densely populated big cities in the east of the country are non-attainment, while the more open country in the west has more attainment areas. Denver is an anomaly because it sits in a valley, so the ozone doesn't rise like it does in other places.
3. Particulate matter has recently been added to the requirements of the Clean Air Act. Theorize reasons that it was not part of the act until the 1990’s.
With industrialization becoming more prominent as time goes on, particulate matter has become more and more of a problem. It eventually became much more noticeable. Also, with the new century, people are finding more ways to combat particulate matter, like driving electric cars. This makes it easier to eradicate particulate matter and more plausible to include it in the Clean Air Act now that there are ways to avoid particulate matter.
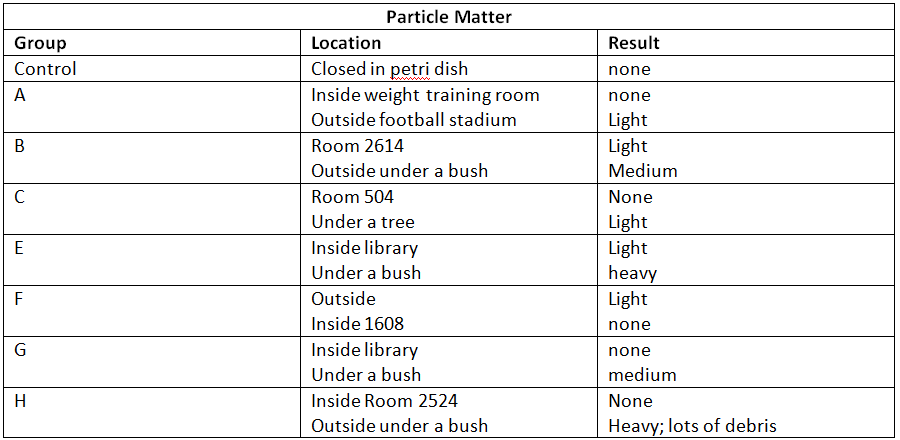
4. Compare and contrast the different PM samples – did they meet your expectations? Why or why not?
The indoor PM samples were light to none while the outdoor samples had light to heavy results. This makes sense because there are more particulates outside in the open air, where everything is exposed, than there are inside. Plant activity, exhaust fumes, smoking, and general dirt outside can help to create particulate matter, but none of that goes on inside. They did meet my expectations because I was sure there would be more PM outside than inside.
5. Explain why air quality monitoring is important for the health of the economy.
Monitoring air quality is important for the health of the economy because poor air quality can damage lungs and exacerbate and cause respiratory disorders. It can induce asthma attacks, which can be fatal. It can also harm unborn children if pregnant mothers are exposed to it, as scientists investigated recently in Sweden (Prime). If we keep the air clean, we keep the people healthy and living longer. Otherwise, harmful conditions can occur that plague and kill humans.
With industrialization becoming more prominent as time goes on, particulate matter has become more and more of a problem. It eventually became much more noticeable. Also, with the new century, people are finding more ways to combat particulate matter, like driving electric cars. This makes it easier to eradicate particulate matter and more plausible to include it in the Clean Air Act now that there are ways to avoid particulate matter.
4. Compare and contrast the different PM samples – did they meet your expectations? Why or why not?
The indoor PM samples were light to none while the outdoor samples had light to heavy results. This makes sense because there are more particulates outside in the open air, where everything is exposed, than there are inside. Plant activity, exhaust fumes, smoking, and general dirt outside can help to create particulate matter, but none of that goes on inside. They did meet my expectations because I was sure there would be more PM outside than inside.
5. Explain why air quality monitoring is important for the health of the economy.
Monitoring air quality is important for the health of the economy because poor air quality can damage lungs and exacerbate and cause respiratory disorders. It can induce asthma attacks, which can be fatal. It can also harm unborn children if pregnant mothers are exposed to it, as scientists investigated recently in Sweden (Prime). If we keep the air clean, we keep the people healthy and living longer. Otherwise, harmful conditions can occur that plague and kill humans.
The experiment proved that ground level ozone is more prominent outside than inside, and that while Wake Forest is under the EPA's limit, it still has a high concentration of ozone. This should be monitored and reduced at every opportunity for the safety of others. My hypothesis that there would be more ground level ozone outside than inside was proved correct by the data, which, while it was partially incomplete, shows that ozone strips left outside collected more ozone than those left inside. As for particulate matter, it was all heavier outside than inside.
Ground level ozone is a serious problem that affects people in their everyday lives. This experiment proves that it can be dangerous for some people to even go outside. The Clean Air Act is an important step in the right direction, but there are still too many non-attainment zones to be safe. More classrooms should do experiments like this to open students' eyes to the dangers of poor air quality.
Ground level ozone is a serious problem that affects people in their everyday lives. This experiment proves that it can be dangerous for some people to even go outside. The Clean Air Act is an important step in the right direction, but there are still too many non-attainment zones to be safe. More classrooms should do experiments like this to open students' eyes to the dangers of poor air quality.
Works Cited
Prime, Louise. "Ozone Link to One in 20 Pre-eclampsia Cases." OnMedica. N.p., n.d. Web. 07 Feb. 2013.
Prime, Louise. "Ozone Link to One in 20 Pre-eclampsia Cases." OnMedica. N.p., n.d. Web. 07 Feb. 2013.